What is Tungstic Acid

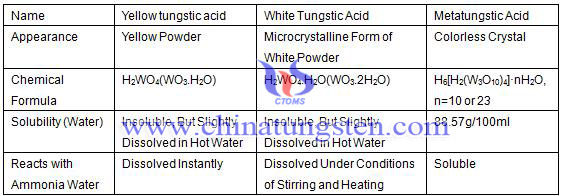
What is tungstic acid? Tungstic acid is the generic term of all kinds of hydrates of tungsten oxide (WO3), polymer compound which formed by the combination of tungsten trioxide WO3 and then united with water at different ratios and formations. The known forms of tungstic acid are yellow, white tungstic acid and metatungstic acid, wherein the yellow tungstic acid is the representative with the basic information like follows:
Product Name: Tungstic Acid or Yellow Tungstic Acid
Chemical Formula: H2WO4
Monocular Mass: 249.86
CAS Number: 7783-03-1
Appearance: Yellow powder
Density: 5.59g/cm3
Solubility (Water): insoluble, but slightly dissolved in hot water
Solubility (Other solvent): dissolved in alkali and ammonia water, and hardly dissolved in inorganic acid except for hydrofluoric acid.
HS Code: 2825901100
Boiling Point: 1473℃
Melting Point: Tungstic acid will lose one molecule of water and become tungsten acid anhydride (Tungsten trioxide) when heated to 100℃.
Application: Mainly used for manufacturing metal tungsten, tungsten wire, tungsten carbide, tungstate ect., and also can be used as printing and dyeing auxiliaries, catalysts and so on.
Contrast table of physical and chemical properties of tungsten acid:
.







